FMEA/Control Charts/Quality Tools
FMEA/Control Charts/Quality Tools


What is the Production Part Approval Process (PPAP)?
PPAP is a design and manufacturing process used to verify a supplier’s ability to reliably and repeatedly produce a part or set of parts for mass production.
The PPAP process consists of 18 steps and the quality tools in Total Lean Management (TLM) software address these steps as shown below in the overall PPAP process.
Organize Early, Grow Faster, Comply Without Compromise
FMEA/ /Control Charts/ Quality Tools
| PPAP Step | TLM Module / Tools |
|---|---|
Design Records: A copy of the drawing or model provided by the customer. | Document Control |
Engineering Change Documents: Detailed description of changes of parts from previous revisions called Engineering Change Notice. | ECR Module Document Control |
Customer Engineering Approval: Customer approval of sample production parts. | Inventory/Parts Attachments |
Customer Engineering Approval: Customer approval of sample production parts. | Inventory/Parts Attachments |
Design Failure Mode and Effects Analysis (DFMEA): Prediction of a product's potential design failure. | FMEA |
Process Flow Diagrams: All steps in the manufacturing process including components, measurement, and inspection. | Document Control |
Process Failure Mode and Effects Analysis (PFMEA): Prediction of a potential process failure that could occur during production. | FMEA |
Control Plan: Details the plan for how quality will be implemented to ensure a stable and reliable process. | Control Plans |
Measurement System Analysis (MSA): Conformance to customer’s ISO or TS standard. Usually Gage R&R for critical impact characteristics to control repeatability and reproducibility and confirmation that gages are calibrated to measure these characteristics to control measurement bias. | Equipment |
Dimensional Results: A list of every dimension noted on the ballooned drawing or model with pass/fail assessment. | Control Plan / Action Plan Checklist |
Material / Performance Test Results: Summary of every test performed on the part, usually in the form of DVP&R (Design Verification Plan and Report). | Document Control |
Initial Process Studies: Shows that critical processes are reliable. Includes SPC (statistical process control) charts | Inspections Metrics |
Qualified Laboratory Documentation: All industry certifications for validation testing. | Equipment / Attachments |
Appearance Approval Report (AAR): Customer approval on final product appearance including color, texture, fit, and more. | Inventory / Attachments |
Sample Product: Sample from initial production run. | N/A |
Master Sample: Sample part signed off by customer and supplier. | Inventory / Attachments |
Checking Aids: Detailed list of all tools used to inspect and measure parts. | Control Plans / Reports / TLM Checklist |
Records of Compliance with Customer-Specific Requirements: List of customer’s specific requirements for PPAP process. | Inventory / Attachments |
Part Submission Warrant (PSW): Summary of entire PPAP submission. | ECR / Projects |
FMEA - Failure Mode Effect Analysis
The FMEA Software Module in TLM allows Fusion Technology™ linking with various other TLM modules in order to accelerate the FMEA process by making access to related information seamless and easy. These links include:
- Parts
- Documents
- Attachments
- Projects
- Deviations Events
- Action Plans
- Sales Orders
- Surveys
The Risk Priority Number (RPN) is a standard FMEA Software risk analysis measurement that can be based on a 3 value or 2 value equation.
3 value: RPN = Severity x Likelihood x Detection
2 value: RPN = Severity x Likelihood
TLM supports both RPN types as the Management Rep can decide which one to use in system settings.
FMEA
- Customizable, color coded, automatically calculated RPN options.
- Establish team members, and their area of responsibility during the FMEA project.
- Link Actions from the analysis to either Corrective Actions or Document Change Requests.
- Create Master project Templates for repeating projects.
- Define and assign events tasks with Gantt chart progress reporting.
- Maintain a Communication Log of Task related messages.
What is a Control Plan?
The Control Plan is a document that describes the actions (measurements, inspections, quality checks or monitoring of process parameters) required at each phase of a process to assure the process outputs will conform to predetermined requirements.
Normally engineers must do this on a spreadsheet without any real data entry help from other data sources, nor can they electronically capture approval signatures that are an important part of the design process.
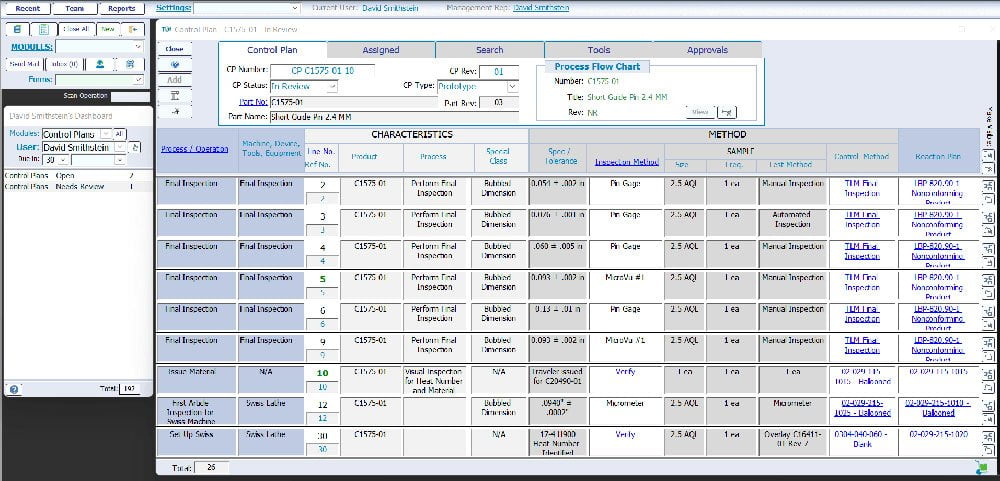
TLM Software makes Control Plans Quick and Easy by
- Leveraging connections with other data sources, such as equipment, work centers, procedures, and Work Order Operations.
- Providing TLM Dashboard support for getting approvals and finding Control Plans in Progress.
- Linking your Work Order Operations with their corresponding Control Plan line item and TLM will automatically update reference numbers if they are changed as new lines are needed, or if the order of work order operations change.
- Review and approval of the control plan can include both employees and/or customers/vendors.
Easy Navigation with Fusion Technology Integration
Fusion Technology is what happens when information integration creates a higher-level management system beyond individual workflows. TLM FMEA/Control Charts/Quality Tools software incorporates this technology by integrating with other TLM solutions to create a Total Lean Management Software approach that provides all stakeholders with a picture of inter-related QMS Software events across your entire organization.
It also serves to create the foundation of your company’s digital landscape which saves you as much as possible from purchasing disconnected stand alone software, and provides the technical support for integrating other information systems when necessary.
Nothing Falls through the Cracks
All TLM software modules are rolled up to a user-specific dashboard and summaries of each user’s dashboard can be automatically emailed to ensure QMS activities are scheduled and prioritized accordingly.
In addition, the TLM dashboard helps users navigate to where tasks are performed, making it easy for new users to adopt the software without having to pay for extra training before being able to put TLM software to work for your company.
Why Do I Need FMEA, Control Charts, and Quality Tools?
FMEA, Control Charts, and Quality Tools within your quality management system software provides your engineers the tools they need to get it right the first time and avoid all the costs and risks associated with less than optimized designs and production processes.
Stabilizes Your Processes: One of the key requirements of understanding the difference between the current state and future state of a process needing improvement is knowing that when the existing process is performed, it’s performed the same way by everyone.
- Do it Right the First Time: FMEA, Control Charts, and Quality Tools provide the level of risk management needed with your quality management system (QMS) software to ensure steps are not forgotten or skipped.
- Managing Risk: FMEA, Control Charts, and Quality Tools can provide important reminders where needed to consider risks involved with a particular process. This simple, two second reminder to consider risks that might be triggered as a result of a given process could one day save the company millions when disaster was averted because someone stopped and asked themselves, “what risks might be triggered here.
Regardless if you are using TLM as QMS software, ERP software, Risk Management Software, Vendor Management software, or IT Risk Analysis software, incorporating Events, Tasks, Action Plans, TLM Checklists or FMEA, Control Charts, and Quality Tools where needed throughout your workflows will have a positive ripple effect on the ability of those procedures or work instructions to get the job right the first time.
Motivate Employees to Excel
Of all the factors that drive human behavior, whether positive or negative, none are as universal as the need to evolve, whether we are conscious of it or not.
Your management team can create impressive organizational growth by harnessing this basic element of the human experience and the FMEA, Control Charts, and Quality Tools in TLM are some of key tools that will make your QMS software successful.
By organizing key aspects of how work is done, managers can use FMEA, Control Charts, and Quality Tools to empower employees to participate in finding opportunities for improvement and customer satisfaction while maintaining approval control to ensure additional risks aren’t introduced.
FMEA, Control Charts, and Quality Tools ensure these efforts are well organized, linked to all other aspects of the QMS software that might be related, and assigned to ensure employees who rise to the occasion can be recognized accordingly, and are motivated to do so as a result.
Improve Every Aspect of Your Company
Historically the decision to implement ISO was driven by the consequences of NOT being ISO certified when competitors were using ISO certification to win new business from customers who needed confidence in their supplier’s ability to consistently meet requirements.
This resulted in a lot of paper based systems set up by various “consultants” that advocated vague procedures that could technically meet the requirements of the ISO standard, but in reality did very little to improve quality or any other aspect of the business.
Times have changed, and the introduction of QMS software allowed organizations to take a different approach to their quality system and instead of wasting tons of time and money for little benefit, they are using their QMS to improve all aspects of the business.
At least the companies who could afford the high cost of most QMS software.
The whole point of ISO based quality standards is to address the minimum risks across every aspect of the business that could have consequences to customer satisfaction. Unfortunately, by calling them “quality” standards, a misperception developed over the years that caused managers to think of quality as something the “quality department” was responsible for.
Total Lean Management (TLM) Software corrects this misperception by acting as an integrated management tool not only for compliance but as the framework for how a company evolves. Audit management software can be a vital component of this evolution, provided it is integrated with all the other aspects of the business to make auditing easier, faster, and more insightful.
There is Only One Type of Quality Management System
Yours!
Over the last 20 years TLM has discovered a very basic truth when it comes to information management systems such as your eQMS, no two companies have the exact same needs.
In fact, the two biggest risks in the software selection process is either selecting software that is not flexible enough to manage information that is unique, and important to your workflow, thus forcing work arounds, or other concessions that effectively add waste to your business.
The other mistake is getting stuck with comprehensive, but inflexible quality management solutions due to their web based cloud hosting design, which forces a one size fits all approach to meeting customer needs.
And while one size might fit some, it might not fit all your needs when in the long run your understanding of those needs evolves and improves.
Total Lean Management Software, by definition, recognizes this fact, and addresses quality management solutions through a dual technology, upstream / downstream user landscape approach to meeting the needs of both your upstream and downstream user types. This is combined with our tiered approach to addressing newly discovered customer needs, making sure the software works in alignment with your business systems the day after your purchase, and for all the years ahead.
TLM Workflow Support:
Custom fields
Allows you to adapt the software to your company’s terminology.
System Settings
Allows you to choose which features are applied to your process.
Rapid update cycles
Incorporating employee feedback into the continuous improvement cycle creates a synergistic effect on moral, engagement, and productivity.
Development Projects
This capability provides insurance that your system will never leave you stuck after discovering unknown needs after implementation.
High levels of interlinking
By implementing and combining modules in way that might be unique to your business or work flows.
World class customer support
Evolution is a team effort, and the whole team behind TLM enjoys helping companies comply with regulations, manage risk, continuously improve, and maintain a steady growth rate.
Help Files can be Linked to your Controlled Work Instructions
This unique TLM feature allows you to link individual screens in TLM to your own controlled procedures or work instructions for that area of the software (if needed)..
Why TLM is your Best QMS Software Choice
TLM Delivers Your Solution
Out of the box, or with a few customizations, our QMS Software is backed by a 100% success rate for over 2 decades getting companies ISO certified on the first try!
If you have any questions or want more detailed information, do not hesitate to call us (866) 209-6896.
“TLM Customers are always our top priority” -David Smithstein, CEO
“In the 10 years that we have relied on TLM to manage our quality documents, it has grown to become the core of our QMS. Our QMS has been audited by BSI, TUV, SAI, FDA, Canada Health and CDPH . We survived all of those audits and emerged a better company. Thanks, David!”

Jim Klett
“EXCELLENT customer service! Without fail, David has responded to every question, email and phone call within a few hours. He is great to work with and always willing to help… TLM is a one-stop shop for all of our QMS needs … The training videos on the website are helpful- keep them coming!”

Allison Getz
“Very helpful in fine-tuning the software/reports to meet our specific needs for audits, even ones requested the day before the audit. The software is straight forward for use, we have 8 personnel in two different locations using it seamlessly … The best customer support I have ever used.”

Stewart Denham
Quality Integrated with Efficiency














 Demos
Demos